One in nine

Ask any woman which disease she fears most and there is a good chance she will say breast cancer. By the time you reach your thirties, or certainly your forties, you will probably know someone who has, or has had breast cancer. It’s the most common cancer affecting women in the UK. Each year, there are over 40,000 new cases. One in every nine women in the UK will develop breast cancer at some point in their lives. Although less prevalent, breast cancer occurs in men too, affecting one in 300 men during their lifetime.
It has been widely publicised that the chances of surviving breast cancer have greatly improved. Not so well-publicised is that the chance of developing breast cancer is a good deal higher too than it was in the 1970s. Between 1971 and 2003, the incidence of breast cancer has increased by a staggering 80 per cent. However, the total number of women dying from breast cancer has remained fairly constant.
A number of potential causative risk factors are generally accepted including the early onset of periods, late menopause, having a first child late in life and, of course, genetics. Many people think that their risk of developing breast cancer is beyond their control, that “fate” will decide. This type of genetic fatalism results from the much-publicised link between genes and breast cancer. However, less than 10 per cent of all breast cancers are caused by faulty genes, the vast majority are caused by environmental factors including diet. Despite a growing body of scientific evidence, there is a widespread reluctance to acknowledge the role of diet in breast cancer risk.
Numerous studies link the consumption of red meat to breast cancer. A range of mechanisms by which red meat leads to an increased risk of breast cancer have been proposed including the presence of carcinogens in meat, the hormone treatment of beef cattle (banned in Europe but practiced widely in the US) or a high intake of haem iron (a highly bioavailable form of iron from meat) which can induce tumour growth. Other studies suggest animal fat may be involved as it can raise steroid hormone levels, and high hormone levels are linked to an increased risk of breast cancer.
The role of hormones and growth factors present in cow’s milk is also a growing concern for many scientists. The hormone content of milk is very different from that of milk produced 100 years ago as modern dairy cows (including organically farmed cows) are frequently impregnated while still producing milk. In fact, two-thirds of retail milk in the UK is taken from pregnant cows, when the hormone content of the milk is markedly elevated. Cow’s milk has been shown to contain over 35 different hormones and 11 growth factors. A recent survey of the published data on the occurrence of hormones in cow’s milk and milk products argues that, in the light of modern dairy farming methods, there is a need to update the data concerning hormonal levels in milk and milk products.
Other scientists are concerned about another bioactive molecule found in milk; a growth factor called insulin-like growth factor 1 (IGF-1). This growth factor sends a strong signal in breast milk (including human and cow’s) from mother to infant directing fast growth and development. IGF-1 is not destroyed during pasteurisation and may be absorbed across the gut wall into the bloodstream. Higher circulating IGF-1 levels are associated with an increased risk of breast cancer and other cancers.
This is undoubtedly difficult for many people to accept as it is deeply entrenched in the British psyche that cow’s milk is a natural and normal drink for people of all ages. However, most people in the world don’t drink milk after weaning; they are lactose intolerant. This means they cannot digest the sugar in milk (lactose). For lactose to be digested, it must be broken down in the intestine by the enzyme lactase to its component monosaccharides glucose and galactose. Glucose can then supply energy to the young animal. All young mammals possess the enzyme lactase and can therefore digest lactose, but this ability is lost in most humans after weaning (commonly after the age of two). In global terms lactose intolerance is very common, affecting around 70 per cent of the world’s population. So, contrary to popular belief in the West, not drinking milk beyond weaning is the norm.
There is some good news though. Many studies show how plant-based foods may offer a protective role against breast cancer. Increasing your intake of vegetables (especially salad vegetables) may help. Increasing the amount of (cereal) fibre in the diet may also reduce breast cancer risk by lowering oestrogen levels. Fibre also protects bowel health, slows sugar and fat absorption and lowers cholesterol. Good sources include fruit and vegetables, wholegrains, pulses, nuts and seeds. Meat and dairy foods do not contain any fibre.
It is beyond the scope of this report to attempt to review all the scientific studies investigating the role of diet in breast cancer. However, this report aims to provide a representative sample, including some key studies. Two consistent themes are very apparent throughout the scientific literature: animal-based foods (meat, animal fat and dairy foods) are linked to an increased risk of breast cancer and plant-based foods (fruit and vegetables, especially salad vegetables and fibre), are linked to a lower risk of this disease.
In summary, this report presents a substantial and representative sample of the evidence showing how dietary factors make a significant contribution to the risk of breast cancer.
For more information on what is the healthiest diet for prevention of, and for those who have, breast cancer see the the guide A Fighting Chance. A guide to healthy eating to help prevent and overcome breast cancer. For more information on the detrimental effects of consuming cow’s milk and dairy products see the Viva!Health’s fully-referenced scientific report White Lies.
Breast development
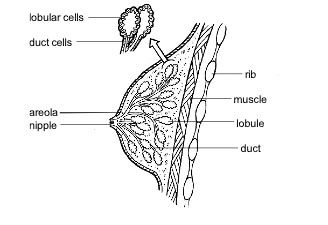 During puberty the sexual organs mature, the secondary sexual characteristics (such as breasts and body hair) develop and reproduction becomes possible. During this time girls develop milk-producing glands called lobules at the back of the breasts. These lobules are connected to tiny tubes called ducts that can carry milk to the nipple. The lobules, ducts and blood vessels are surrounded by fatty tissue and connective tissue called stroma which is attached to the chest wall (see Figure 1.0).
During puberty the sexual organs mature, the secondary sexual characteristics (such as breasts and body hair) develop and reproduction becomes possible. During this time girls develop milk-producing glands called lobules at the back of the breasts. These lobules are connected to tiny tubes called ducts that can carry milk to the nipple. The lobules, ducts and blood vessels are surrounded by fatty tissue and connective tissue called stroma which is attached to the chest wall (see Figure 1.0).
The male breast
Men have much less fatty tissue in their breasts than women but can still be affected by breast cancer. Men’s breast tissue contains ducts, but only a few, if any, lobules.
Lymph nodes
The lymphatic system is an important part of the immune system that helps the body fight infection. It is made up of a network of thin vessels that spread into tissues throughout the body. A clear fluid called lymph circulates around the system transporting infection-fighting cells called lymphocytes that help remove foreign matter and cellular debris. Lymph nodes act as filters and may swell up when a possible threat (such as a bacterium) is identified. Generalised lymphadenopathy (when all the nodes of the body are swollen) may indicate a systemic illness such as an infection or cancer.
Breast Awareness
What to watch for
It is important to know how your breasts look and feel normally so that you can spot any changes as they occur. Early detection of breast cancer increases the chances of effective treatment. There are several changes to watch for:
- If one breast becomes larger than the other
- If a nipple becomes inverted
- Rashes on or around the nipple
- Discharge from one or both nipples
- Skin texture changes (puckering or dimpling)
- Swelling under the armpit or around the collarbone (where the lymph nodes are)
- A lump that you feel is different to the rest of your breast tissue
- Continuous pain in one part of the breast or armpit (not a common symptom)
What to do next
If you do notice one or more of these changes then make an appointment to see your GP as soon as you can but try not to worry too much as most breast symptoms do not turn out to be breast cancer. Non-cancerous or benign breast conditions that may cause breast changes include fibrocystic mastopathy, mastitis and fibroadenoma, to name but a few.
Different types of breast cancer
When breast cancer does occur, cells in part of the breast grow in an uncontrolled way. If the cancer is not treated, the cells can spread within the breast or even further, travelling to other parts of the body. If the cancer cells have not spread, it is called non-invasive breast cancer. If the cancer cells develop in the ducts, the cancer is called ductal carcinoma in situ (DCIS), if they occur in the lobules, it is referred to as lobular carcinoma in situ (LCIS). If the cancer cells have spread (metastasised) into the surrounding breast tissue, lymph glands or further within the body, it is called invasive breast cancer.
Number crunching
Breast cancer is the most common cancer affecting women in the UK and one in nine women will have this disease at some point in their lives (NHS Direct, 2007). Women in the US now have an astonishing one in eight lifetime chance of developing breast cancer (American Cancer Society, 2007). There are over 40,000 new cases in the UK every year, representing a third of all cancers in women. Between 1971 and 2003, the incidence rates of breast cancer have increased by 80 per cent (National Statistics, 2005). In the UK in 2005 there were 12,509 deaths from breast cancer (99 per cent in women, one per cent in men). Breast cancer accounts for 17 per cent of female deaths from cancer in the UK (Cancer Research UK, 2007).
Male breast cancer
Although less prevalent, breast cancer does occur in men too; one in 300 men in the UK will have breast cancer at some point in their lives (NHS Direct, 2007). Furthermore, the incidence of breast cancer among men is increasing. One large-scale study of more than 2,500 American men with the disease showed that between 1973 and 1998 the incidence of breast cancer among men increased by 26 per cent (Giordano et al., 2004). This study found that men tended to have larger tumours which had spread further by the time they sought help. That said, breast cancer remains a rare disease among men. Lung, prostate, bowel, oesophageal and stomach cancer are the five biggest causes of cancer death among men in the UK (Cancer Research UK, 2003).
Breast cancer around the world
Disease incidence rates measure the number of new diagnoses per 100,000 people during a defined time period (usually a given year). Age standardisation, often referred to as ‘age-adjustment’, is used to eliminate the confounding effects of differences in the age composition of different populations. This allows us to make statistical comparisons of incidence rates between different populations.
The age-adjusted incidence rates for breast cancer per 100,000 women differ markedly from one country to another. For example, Uruguay has a very high rate at 114.9, followed by 92.1 in the US and 87.1 in Israel. Much lower rates are seen in Korea at just 12.7, 20.0 in Mali and 16.1 in Thailand (Ganmaa and Sato, 2005). In response to this discrepancy, an increasing amount of attention is now focusing on the links between diet and breast cancer, particularly the relationship between the consumption of animal-based foods (meat and milk) and breast cancer.
Population studies
Studying cancer incidence among particular groups of people can provide useful insights into the causes of disease. Researchers from the London School of Hygiene and Tropical Medicine recently reported breast cancer incidence is substantially lower, and survival rates higher, in South Asians living in the UK than other women (Farooq and Coleman, 2005). The authors of this study suggested that differences in diet and lifestyle could explain the different rates observed.
Earlier research published in the British Journal of Cancer also showed that South Asian women living in the UK are less likely to be diagnosed with breast cancer than other women, but found that the risk varied according to their specific ethnic subgroup. This research showed that Muslim women from India and Pakistan are almost twice as likely to develop breast cancer as Gujarati Hindu women. This study examined the diet and found that the Gujarati Hindu women were more likely to be vegetarian and therefore had more fibre in their diet due to their higher intake of fruit and vegetables (McCormack et al., 2004). There are several mechanisms by which the diet might influence breast cancer risk. One possible mechanism is through an effect on hormones: increasing the amount of fibre in the diet may reduce breast cancer risk by altering the levels of female hormones (oestrogens) circulating in the blood (Gerber, 1998).
Oestrogen sensitive cancers
Oestrogen receptor-positive cancer (also called oestrogen-sensitive cancer) is when there are specific proteins on the cancer cell’s surface that respond to the hormone oestrogen by causing the cell to grow. Oestrogen receptor-positive cancer makes up around 75 per cent of breast cancers in postmenopausal women and around 50 to 60 per cent in premenopausal women (Breast Cancer Care, 2007). Determining whether a breast cancer tumour is positive for oestrogen receptors can help to guide treatment and determine prognosis.
Current national guidelines recommend that women who have oestrogen receptor-positive breast cancer should usually be offered hormone therapy and women with oestrogen receptor-negative breast cancer should be offered chemotherapy (Cancer Research UK, 2002).
The risk of developing breast cancer is very small in young women and increases as women get older; more than half of breast cancers occur in women over the age of 65 (Cancerbackup, 2007). Some factors may slightly increase a woman’s risk of developing the disease, these are listed below:
- Having had breast cancer
- Having benign (non-cancerous) breast disease
- Genetics – breast cancer in the family (see below)
- Early puberty/menstruation – before the age of 11
- Late menopause – after age 54
- Having a first child late in life
- Having no or few children
- Not breast-feeding long term
- Exposure to radiation
- High dietary fat intake
- Overweight and obesity – particularly for postmenopausal women
- Moderate to heavy consumption of alcohol
- Oral contraceptives (the pill) and hormone replacement therapy (HRT) may very slightly increase the risk of breast cancer
In addition to the above, studies have included a small number of chemicals identified as mammary carcinogens or hormone disruptors which may have implications for breast cancer. However, evidence is emerging for associations between breast cancer and polychlorinated biphenyls (PCBs), polycyclic aromatic hydrocarbons (PAHs) and organic solvents (Brody et al., 2007). PCBs are persistent organic pollutants that do not degrade easily and so are widespread in the environment. They are generally present at low concentrations in most foods, especially fat-containing foods such as milk and meat (FSA, 2000). PAHs are ubiquitous in air pollutants produced from vehicle exhausts (Brody et al., 2007). Organic solvents are commonly used in detergents (citrus terpenes), dry cleaning (tetrachloroethylene), paint thinners (toluene and turpentine), nail polish removers and glue solvents (acetone, methyl acetate, ethyl acetate) and in perfumes (ethanol).
Just how much some of these factors contribute to the risk of breast cancer is difficult to say. However, the aim of this report is to investigate the somewhat overlooked role of dietary factors. That said, the role of genetics in breast cancer deserves further discussion.
The genetic link
Much has been made of the link between genes and breast cancer. The genes BRCA1 and BRCA2 have received the most attention since they were first discovered in 1994 and 1995 respectively. A fault in either of these two genes can increase the chances of getting breast cancer. However, even among women with high-risk BRCA1 and BRCA2 gene mutations, evidence suggests that nongenetic (environmental) factors influence risk. A substantial review of the research, published in the journal Cancer, reported that more women born after 1940, carrying a fault in either, or both of these genes, were diagnosed with breast cancer by the age of 50 than those born before 1940 (Brody et al., 2007). In other words, the women born after 1940 were exposed to some environmental factor that increased their risk of developing breast cancer.
There are two other very rare genes which are thought to account for less than two per cent of all breast cancers: the P53 gene and the AT (ataxia telangiectasia) gene (Cancerbackup, 2006).
It is important to remember that having an abnormal gene does not mean that a person will definitely develop breast cancer, but does mean they are considerably more at risk of developing the condition than someone who does not have one of the abnormal genes. Interestingly, with a faulty gene, the probability of developing cancer has increased over the last 30 or more years from about 40 per cent to about 70 per cent, probably due to environmental and lifestyle factors (CANCERactive, 2007). In other words you can cut your risks, even if you have a faulty gene, by making changes to your diet and lifestyle.
The discovery of genes linked to breast cancer has given rise to a certain degree of ‘genetic fatalism’, encouraging some to think that their chances of getting breast cancer are entirely down to fate. However, scientists estimate that only around five to 10 per cent of breast cancers are due to inheriting abnormal genes (Cancerbackup, 2007). This means that the vast majority of cancers (90 per cent) are not caused by abnormal genes. Secondly, it is important to remember that having an abnormal gene does not mean that a person will definitely develop breast cancer, but does mean they are more at risk of developing the condition than someone who does not have one of the abnormal genes.
Dietary Factors
An increasing body of scientific evidence links certain foods (such as meat and dairy products) to an increased risk of breast cancer. Conversely, other studies have identified foods that may lower the risk. As communications technology advances, scientists have become more able to compare disease rates and dietary patterns around the world. This has afforded them some useful incites into the causes of disease.
East versus West
Cross-cultural studies show that as the consumption of a typical Western diet (containing high levels of saturated fat, cholesterol and animal protein) spreads from country to country, so does the incidence of the so-called ‘diseases of affluence’ (such as obesity, heart disease, diabetes, osteoporosis and certain cancers, including breast cancer). It has been suggested that the incidence of these diseases varies because of genetic differences between different races. However, when people migrate from an area of low incidence of the so-called affluent diseases to an area of high incidence, they soon acquire the same high incidence shared by the population into which they have moved. This correlation must then be attributed, at least in part, to environmental factors such as diet and lifestyle.
So if certain diets and lifestyles increase the risk of these diseases, it stands to reason that you can reduce the risk of disease by changing your diet and lifestyle.
As the typical Western diet pervades around the world, it takes with it typical Western diseases such as heart disease, diabetes, obesity and certain cancers. The World Health Organisation (WHO) states that dietary factors are estimated to account for around 30 per cent of all cancers in Western countries and 20 per cent of cancers in developing countries. They predict the number of cancers linked to diet in developing countries will grow as these countries become more urbanised (WHO, 2007). Other estimates are even higher. In 1997, the World Cancer Research Fund published a substantial review of the scientific literature linking foods, nutrition, food processing, dietary patterns and related factors, with the risk of human cancers worldwide (WCRF, 1997). The report concluded that policy-makers should now recognise that the incidence of cancer throughout the world can be reduced by 30 to 40 per cent by feasible changes in diets and related lifestyles.
Sadly there has never been a better time to observe the detrimental effects of the Western diet as countries in the East (such as China and Japan) move from a traditional plant-based diet rich in fruit, vegetables and grains towards a more Western diet characterised by meat, dairy and processed foods.
In a recent study, the effects of a Western diet on breast cancer risk was assessed among participants of the Shanghai Breast Cancer Study, a large population-based survey involving 1,446 Chinese women from Shanghai diagnosed with breast cancer and matched to 1,549 control subjects without the disease (Cui et al., 2007). The authors of the report identified two distinct dietary patterns which they describe as “vegetable-soy” including a high intake of vegetable, soya products and fish and the Western-style “meat-sweet” diet characterised by various meats, poultry, fish, confectionary, puddings, bread and milk. The “vegetable-soy” diet contained higher levels of fibre, vitamins C and E and soya protein while the “meat-sweet” diet was rich in total and saturated fat.
Results showed no overall association of breast cancer risk with the “vegetable-soy” pattern but demonstrated that a Western-style diet could double the rate of breast cancer among postmenopausal overweight women. Previous work has suggested that a “vegetable-soy” type diet can lower the risk of breast cancer. However, the authors of this study found that the “vegetable-soy” diet did not protect from breast cancer. This, they suggested, might due to the negative effects of the fish content of the “vegetable-soy” diet, or that cooking the vegetables may have diminished their protective effects (see below). The authors of this study concluded that for postmenopausal women in traditionally low-risk Asian populations poised to adopt foods characteristics of Western societies, the low consumption of a “meat-sweet” dietary pattern plus successful weight control may protect against breast cancer.
Red meat
While previous work has shown that red meat increases the risk of bowel, stomach and pancreatic cancer, recent research now links red meat to breast cancer as well (Cho et al., 2006). Researchers from Harvard Medical School in Boston looked at the diets of over 90,000 premenopausal women (aged 26 to 46) over 12 years and found that women who ate more than one-and-a-half servings of red meat per day (the equivalent of a sausage and a burger) almost doubled their risk of oestrogen receptor-positive breast cancer. The authors of this study concluded that a high red meat intake may be a risk factor for breast cancer.
They suggest several biological mechanisms that may explain how this occurs. For example, cooked or processed red meat is a source of carcinogens (cancer-causing agents such as heterocyclic amines, N nitroso-compounds, and polycyclic aromatic hydrocarbons), that may increase breast cancer risk. Hormone treatment of beef cattle for growth promotion (banned in European countries but not in the United States) is also a concern. Red meat is a source of haem iron (a highly bioavailable form of iron and a major source of stored body iron), which has been shown to enhance oestrogen-induced tumour induction. Finally, they state that fat intake in general has been hypothesised to raise steroid hormone levels (see below). However, in a previous study, this research group found that intake of animal fat, but not vegetable fat, was related to elevated risk of breast cancer (Cho et al., 2003).
Exactly how red meat may increase the risk of breast cancer remains unclear. This uncertainty inevitably casts some doubt on the role of red meat as a causative agent, particularly among enthusiastic meat-eaters. However, the mechanism by which red meat causes bowel cancer was only just proposed recently (Lewin et al., 2006) despite its causal role being suspected for many decades. In other words, we know that increasing consumption of red meat increases the risk of developing breast cancer, but scientists do not yet know exactly how.
Animal fat
Some studies link dietary fat to the risk of breast cancer. Case-control studies use a group of people with a particular characteristic (for example older women with lung cancer). This particular group is selected and information collected (for example, history of smoking), then a control group is selected from a similar population (older women without lung cancer) to see if they smoked or not, then a conclusion is drawn (smoking does or does not increase risk of lung cancer). A combined analysis of 12 case-control studies designed to examine diet and breast cancer risk found a positive association between fat intake and this disease. It was concluded that the percentage of breast cancers that might be prevented by dietary modification in the North American population was 24 per cent for postmenopausal women and 16 per cent for premenopausal women (Howe et al., 1990).
In 1999 researchers at the Department of Preventive Medicine at the University of Southern California Medical School in Los Angeles published a review of 13 dietary fat intervention studies that were conducted to investigate the effect of fat intake on oestrogen levels. The results showed decreasing dietary fat intake (to between 10 and 25 per cent of the total energy intake) reduced serum oestradiol levels by between 2.7 and 10.3 per cent. It was concluded that dietary fat reduction can result in a lowering of serum oestradiol levels and that such a dietary modification may help prevent breast cancer (Wu et al., 1999).
However, other studies of fat intake and the incidence of breast cancer have yielded conflicting results. The discrepancy in results may reflect the difficulties of accurately recording fat intake. Dr Sheila Bingham of the Dunn Human Nutrition Unit in Cambridge has developed a data-collection method which may overcome these problems. Bingham used food frequency questionnaire methods with a detailed seven-day food diary in over 13,000 women between 1993 and 1997. The study concluded that those who ate the most animal saturated fat (found mainly in whole milk, butter, meat, cakes and biscuits) were almost twice as likely to develop breast cancer as those who ate the least. It was also concluded that previous studies may have failed to establish this link because of imprecise methods (Bingham et al., 2003).
In a subsequent prospective cohort study involving over 90,000 premenopausal women, researchers from Harvard Medical School confirmed that animal fat intake was associated with an elevated risk of breast cancer (Cho et al., 2003). Red meat and high-fat dairy foods such as whole milk, cream, ice-cream, butter, cream cheese and cheese were the major contributors of animal fat in this cohort of relatively young women. Interestingly, this research did not find any clear association between vegetable fat and breast cancer risk; the increased risk was only associated with animal fat intake. It has been suggested that a high-fat diet increases the risk of breast cancer by elevating concentrations of oestrogen. However, the authors of this study suggest that if this were true a diet high in animal fat and a diet high in vegetable fat should both lead to higher rates of cancer, and that was not the case in this study. They do state that the fatty acid composition in fat from animal and vegetable sources differs greatly and may therefore have different associations with breast cancer risk. So it could be that a high-fat plant-based diet has less effect than a high-fat diet containing lots of meat and dairy products. They also suggest that some other component such as the hormones in cow’s milk might play a role in increasing the risk of breast cancer.
Oestrogen
The conflicting effects of animal and vegetable fats on breast cancer risk have led many research groups to focus on the endogenous (naturally produced) hormonal content of animal-based foods, which has not been widely discussed. Oestrogens are contained in meat and eggs, but the major source of animal-derived oestrogens in the human diet are cow’s milk and dairy products which account for 60 to 80 per cent of the animal-derived oestrogens consumed (Hartmann et al., 1998).
Furthermore, the milk produced now is very different from that produced 100 years ago; modern dairy cows are frequently impregnated while still producing milk (Webster, 2005). Two-thirds of milk in the UK is taken from pregnant cows with the remainder coming from cows that have recently given birth. This means that the hormone (oestrogen, progesterone and androgen precursor) content of milk varies widely. It is the high levels of hormones in animal-based foods that have been linked to the development of hormone-dependent cancers such as breast cancer.
Numerous studies show that women consuming a Western-style diet tend to have different hormone profiles compared to women eating traditional diets. This means the level of oestrogen in their blood varies according to the type of diet they have. For example, a review of studies carried out over a 10 year period in the Department of Clinical Chemistry at the University of Helsinki in Finland showed that women who consume a high-animal fat, high-animal protein diet with mostly refined carbohydrates and sugars have higher levels of oestrogen in their blood (Adlercreutz, 1990). This study also made the observation that the hormone pattern (high oestrogen), found in association with a Western-type diet tends to prevail among breast cancer patients. In other words, what you eat can affect the level of oestrogen in the blood, and high oestrogen levels are found in women with breast cancer.
A number of other studies confirm that women with breast cancer tend to have higher levels of circulating oestrogens. Prospective studies follow groups of people over time. Generally these people are alike in many, but not all, ways (for example, young women who smoke and young women who do not). The prospective cohort study will then look for a link between their behaviour and a particular outcome (such as lung cancer). A prospective study conducted on the island of Guernsey examined serum levels of the oestrogen hormone oestradiol in samples taken from 61 postmenopausal women who developed breast cancer an average of 7.8 years after blood collection (Thomas et al., 1997). Compared to 179 age-matched controls, oestradiol levels were 29 per cent higher in women who later developed breast cancer.
Another prospective study (this time from the US) compared oestrogen levels in 156 postmenopausal women who developed breast cancer, after blood collection, with two age-matched controls for each cancer patient (Hankinson et al., 1998). Results showed increased levels of the hormones oestradiol, oestrone, oestrone sulphate and dehydroepiandrosterone sulphate in women
who subsequently developed breast cancer thus providing strong evidence for a causal relationship between postmenopausal oestrogen levels and the risk of breast cancer.
In a review looking at the relationship between breast cancer incidence and food intake among the populations of 40 different countries, a positive correlation was seen between the consumption of meat, milk and cheese and the incidence of breast (and ovarian) cancer (Ganmaa and Sato, 2005). Meat was most closely correlated with breast cancer incidence, followed by cow’s milk and cheese. By contrast, cereals and pulses were negatively correlated with the incidence of breast cancer. The authors of this review concluded that the increased consumption of animal foods may increase the development of hormone-dependent cancers. Among dietary risk factors of particular concern to the authors were milk and dairy products, because so much of the milk we drink today is produced from pregnant cows, in which hormone levels are markedly elevated.
Milk contains many biologically active (bioactive) molecules including enzymes, hormones and growth factors. In 1992, Pennsylvania State University endocrinologist Clark Grosvenor published an extensive review of some of the known bioactive hormones and growth factors found in a typical glass of milk in the US. The list included seven pituitary (an endocrine gland in the brain) hormones, seven steroid hormones, seven hypothalamic (another brain endocrine gland) hormones, eight gastrointestinal peptides (chains of two or more amino acids), six thyroid and parathyroid hormones, 11 growth factors, and nine other biologically active compounds (Grosvenor et al., 1992).
A more recent survey of the published data on the occurrence of hormones and bioactive constituents in cow’s milk and milk products also provides an extensive list of gonadal, adrenal, pituitary, hypothalamic and other hormones (Jouan et al., 2006). The authors of this survey state that there is a need to update the data concerning hormonal levels in milk and milk products, especially in the light of changes in the genetic background of dairy cattle in the last decades, as well as in animal feeding and husbandry and new processes that have emerged in the milk industry (Jouan et al., 2006).
The enormous detrimental health effects of consuming cow’s milk and dairy products are more widely discussed in the VVF’s fully-referenced scientific report White Lies. This report describes how the saturated animal fat, animal protein, cholesterol, hormones and growth factors in dairy products are linked to a wide range of illnesses and diseases including some of the UK’s biggest killers such as heart disease, diabetes, prostate cancer as well as osteoporosis, eczema, asthma, Crohn’s disease, colic, constipation and even teenage acne.
For more information on British dairy farming methods see Viva!’s fully-referenced report The Dark Side of Dairy.
IGF-1 signalling trouble
In addition to the animal fat, various chemical contaminants and hormones found in animal-based foods, certain growth factors have been implicated in the proliferation of human breast cancer cells. In particular, a growth factor called insulin-like growth factor 1 (IGF-1) has attracted much attention.
IGF-1 is a signalling molecule produced in the liver and body tissues of mammals. It promotes cell growth and division, which is important for normal growth and development of mammals. However, IGF-1 levels decline with age, consistent with their role in growth.
Over the last decade IGF-1 has been linked to an increased risk of childhood cancers, breast cancer, lung cancer, melanoma and cancers of the pancreas and prostate (LeRoith et al., 1995; Chan et al., 1998) and gastrointestinal cancers (Epstein, 1996). Indeed IGF-1 may be used as a predictor of certain cancers, in much the same way that cholesterol is a predictor of heart disease (Campbell and Campbell, 2005).
IGF-1 from cows is identical to human IGF-1 in that the amino acid sequence of both molecules is the same (Honegger and Humbel, 1986). Amino acids are the building blocks of proteins and there are 20 different amino acids. All proteins consist of amino acids joined together like beads on a string and the nature of the protein (how it behaves) is determined by the order in which the amino acids occur along the string. In both human and bovine IGF-1 the same 70 amino acids occur in exactly the same order, which would suggest that bovine IGF-1 behaves the same way in humans as it does in cows.
As stated above, two-thirds of milk in the UK is taken from pregnant cows. It is not only the hormone content that is markedly elevated at this time; the amount of IGF-1 present is also higher in milk produced by pregnant cows. IGF-1 is relatively stable to both heat and acidic conditions and can therefore survive the harsh conditions of both commercial milk processing and gastric acid to maintain its biological activity (Playford et al., 2000). Some scientists are concerned that IGF-1 not destroyed during pasteurisation may cross the intestinal wall in the same way that another hormone, epidermal growth factor (EGF), has been shown to do. EGF is protected from being broken down when food proteins (such as the milk protein casein) block the active sites of the digestive enzymes (Playford et al., 1993). This allows the molecule to stay intact and cross the intestinal wall and enter the blood. Dr J.L. Outwater of the Physicians Committee For Responsible Medicine (PCRM) in Washington, DC, warns regular milk ingestion after weaning may produce enough IGF-1 in mammary tissue to encourage cell division thus increasing the risk of cancer (Outwater et al., 1997).
Furthermore, some research shows that various hormones and growth factors (including oestrogens, adrenocorticotropic hormone, thyrotropin, luteinizing hormone, follicle-stimulating hormone, platelet-derived growth factor, epidermal growth factor and fibroblast growth factor) can affect IGF-1 production within the body (Yu and Rohan, 2000). This indicates that certain foods, such as cow’s milk, may increase endogenous IGF-1 production in humans. So diet can determine the amount of IGF-1 present in the blood.
The critical role IGFs play in regulating cell growth and death has led to much speculation about
their involvement in cancer development (Yu and Rohan, 2000). IGF-1 regulates cell growth, development and division and can stimulate growth in both normal and cancerous cells. Indeed, IGF-1 has been shown to stimulate the growth of human breast cancer cells in the laboratory and it has been suggested that it may be involved in the transformation of normal breast tissue to cancerous cells (Outwater et al., 1997). The concern here is that if IGF-1 can cause human cancer cells to grow in a Petri dish in the laboratory, it might have a cancer-inducing effect when consumed in the diet. This is very worrying as even small increases in serum levels of IGF-1 in humans are associated with increased risk for several common cancers including cancers of the breast, prostate, lung and colon (Wu et al., 2002).
In the first prospective study to investigate the relationship between the risk of breast cancer and circulating IGF-1 levels, researchers at Harvard Medical School analysed blood samples originally collected from 32,826 women aged between 43 and 69 years during 1989 and 1990 (Hankinson et al., 1998a). From this group, 397 women were later diagnosed with breast cancer. Analysis of IGF-1 levels in samples collected from these women compared to samples from 620 controls (without breast cancer) revealed a positive relationship between circulating IGF-1 levels and the risk of breast cancer among premenopausal (but not postmenopausal) women. It was concluded that plasma IGF-1 concentrations may be useful in the identification of women at high risk of breast cancer.
Taken together, the scientific literature strongly suggests a link between high circulating IGF-1 levels and cancer, but what has this to do with diet? The answer is a lot: circulating IGF-1 levels are higher in people who consume milk and dairy products. US researchers from Harvard Medical School investigating the link between IGF-1 levels and diet examined IGF-1 levels in 1,037 healthy women (Holmes et al., 2002). The most consistent finding of this study was a positive association between circulating IGF-1 levels and animal protein intake which, in this study, was largely attributed to cow’s milk intake.
Researchers at the Fred Hutchinson Cancer Research Centre in Washington investigated the link between plasma levels of IGF-1 and lifestyle factors in 333 people (Morimoto et al., 2005). They too found that milk consumption was linked to IGF-1 levels. This concurs with an earlier study, from Creighton University in Omaha, NE, which observed a 10 per cent increase in blood levels of IGF-1 in subjects who increased their intake of non-fat milk from fewer than 1.5 servings of dairy foods to three servings per day (Heaney, 1999). Furthermore, a study from the Cancer Epidemiology Unit at the Radcliffe Infirmary in Oxford noted that vegan men had a nine per cent lower serum IGF-1 level than meat-eaters and vegetarians (Allen et al., 2000). This strongly suggests a link between the consumption of cow’s milk and dairy products and higher IGF-1 levels circulating in the blood.
One study actually quantified the effect of cow’s milk on circulating IGF-1 levels in 54 Danish boys aged two-and-a-half years old (Hoppe et al., 2004). In this study an increase in cow’s milk intake from 200 to 600ml per day corresponded to a massive 30 per cent increase in circulating IGF-1.
Researchers at Bristol University investigating the association of diet with IGF-1 in 344 disease-free men found that raised levels of IGF-1 were associated with higher intakes of milk, dairy products and calcium while lower levels of IGF-1 were associated with high vegetable consumption, particularly tomatoes (Gunnell et al., 2003). In their study, published in the British Journal of Cancer, it was concluded that IGF-1 may mediate some diet-cancer associations.
In conclusion, the research shows that nutrition has an important role in determining serum IGF-1 levels (Yaker et al., 2005). Whether the increase in IGF-1 caused by cow’s milk occurs directly (by IGF-1 crossing the gut wall), or indirectly (as a result of the action of other factors), the evidence suggests that some component of cow’s milk causes an increase in blood serum levels of IGF-1, which in turn is linked to various cancers.
Bovine somatotrophin (BST)
Mammalian milk production is regulated by a complex interaction of hormones. Bovine somatotrophin (BST) is a natural growth hormone that occurs in cattle and controls the amount of milk that they produce. In 1994 Monsanto began marketing a synthetic version of BST, known as recombinant BST (rBST), which was sold as Posilac. Injecting dairy cows with rBST alters their metabolism to increase milk production by up to 15 per cent. Since its introduction in 1994, Posilac has become the largest selling dairy animal pharmaceutical product in the US. Sold in all 50 states, rBST is used in around one-third of the nine million dairy cows in the US (Monsanto, 2007).
While the US Food and Drug Administration (FDA) permit the use of rBST, its use is associated with severe animal welfare problems, for example increasing the incidence of lameness and mastitis. For these reasons, the use of rBST in the EU was prohibited in 2000. Indeed Canada, Japan and many other countries have banned the use of rBST because of its effects on animal health and welfare. However, there are no restrictions on the import of rBST dairy products, or any requirement to label them.
The government’s Veterinary Medicines Directorate does not carry out any testing of imported milk (Defra, 2006). Furthermore, Defra confirmed in correspondence with the VVF that, since the EU is a single market, once a product has entered, if it is transported on to another country within the EU, then the origin of the product will be the EU country rather than the originating country (Defra, 2006). In 2005, the UK imported over 1,000 tons of dairy products (mainly ice-cream) from the US (Defra, 2006a); these figures have declined from over 5,000 tons in both 2001 and 2002 but still remain a concern, especially as the consumer has a limited chance of discriminating against imports from the US. The sensible option is to avoid all dairy products.
Milk production increases in cows treated with rBST because it promotes the production of IGF-1 which then stimulates the glands in the cow’s udders to produce more milk. Research shows that rBST use on dairy cows can substantially increase the levels of IGF-1 in their milk (Prosser et al., 1989). This raises concerns about the potential biological action of IGF-1 from cow’s milk in humans especially because IGF-1 from cows is identical to human IGF-1. Professor Samuel Epstein,
an international leading authority on the causes and prevention of cancer, warns that converging lines of evidence incriminate IGF-1 in rBST milk as a potential risk factor for both breast and gastrointestinal cancers (Epstein, 1996).
So why should this concern us if we do not allow the use of rBST in the UK? Well in terms of human health, the concern is that milk and milk products imported from countries that permit the use of rBST may lead to the consumption of foods that promote increased levels of IGF-1 in humans. In 1999, the minister of state, Baroness Hayman, referred to a report from the Veterinary Products Committee (VPC) which stated that while the use of rBST does not increase the level of BST found naturally in cow’s milk, there is a two-to-five fold increase in the level of IGF-1 in the milk, which she acknowledged may be implicated in the occurrence of colonic cancer. However, Hayman reiterated the VPC’s view that the risk to human health was likely to be extremely small. Hayman also suggested that just 0.3 per cent of total milk and milk products imported into the UK come from the US where rBST is authorised for use (UK Parliament, 1999).
The damaging effects of dairy
In recent years an increasing body of evidence has accumulated linking the consumption of cow’s milk and dairy products to breast cancer. In her book Your Life in Your Hands, Professor Jane Plant CBE, Anglo American Professor of Applied Geochemistry at Imperial College, London, describes a very personal and moving story of how she overcame breast cancer by excluding all dairy products from her diet (Plant, 2007). Plant was diagnosed with breast cancer in 1987. She had five recurrences of the disease and by 1993 the cancer had spread to her lymphatic system. She could feel the lump on her neck, and was told that she had just three months to live, six if she was lucky. However, Plant was determined to use her scientific training to find a solution to this ‘problem’. She began comparing breast cancer incidence in the UK to that in other cultures.
As stated above, the age-standardised breast cancer incidence rate allows the comparison of cancer rates between populations that may have a different age structure. Plant looked at breast cancer incidence rates in rural China where, in 1997, the disease affected 11 out of every 100,000 women (compared to 70 in the UK). This rate was trebled in Chinese cities, probably Plant suggests, because of the pollution and an increased exposure to a more Western style diet. Interestingly, she observes, Hiroshima and Nagasaki have similar rates to those found in Chinese cities. Both places were attacked with nuclear weapons so you might expect to see some radiation-related cancers. However, by moving to Hiroshima and adopting a Japanese lifestyle, Plant reveals, a UK woman would slash her risk of developing breast cancer by half!
Furthermore Plant observed that among wealthy Chinese women with a more Western lifestyle (for example in Malaysia and Singapore), the rate of breast cancer is similar to that in the West. Furthermore, epidemiological evidence shows that when Chinese women move to the West, within one or two generations their rates of breast cancer incidence and mortality increase to match those of their host country. This suggested that diet and lifestyle (rather than genetics) must be a major determinant of cancer risk.
Plant decided to investigate the role of diet in breast cancer risk. She examined the results of the China-Cornell-Oxford project on nutrition, environment and health (Campbell and Junshi, 1994). This project was based on national surveys conducted between 1983 and 1984 in China. The project was a collaboration between T. Colin Campbell at Cornell University in the US, Chen Junshi from the Chinese Academy of Preventative Medicine, in Beijing, China, Li Junyao at the Chinese Academy of Medical Sciences, Beijing, and Richard Peto from Oxford University in the UK. The project revealed some surprising insights into diet and health. For example, it showed that people in China tend to consume more calories per day than people in the US, but only 14 per cent of these calories come from fat compared to a massive 36 per cent in the West. This coupled to the fact that Chinese people tend to be more physically active than people in the West, is why obesity affects far more people in the West than in China.
However, Plant’s diet had not been particularly high in fat; indeed she describes it as very low in fat and high in fibre. Then Plant had a revelation: the Chinese don’t eat dairy produce. Plant had been eating low-fat yogurt and skimmed organic milk up until this time, but within days of ceasing all dairy, the lump on her neck began to shrink. The tumour decreased and eventually disappeared, leading her to the conviction that there is a causal link between the consumption of dairy products and breast cancer. Although Plant received chemotherapy during this time, it did not appear to be working and so convinced was her cancer specialist that it was the change in diet that saved her life, he now refers to cancer mortality maps in his lectures and recommends a dairy-free diet to his breast cancer patients.
Plant eventually defeated cancer by eliminating dairy products from her diet, replacing them with healthy alternatives and making some lifestyle changes. Her book recounts not only her own story but that of others and it contains much research using the peer-reviewed scientific literature. Plant advises that if you do only one thing to cut your risk of breast cancer, make the change from dairy to soya (Plant, 2007). Providing breast cancer patients with sound dietary advice could greatly increase survival rates. Taken together, these observations show that a plant-based diet can reduce many of the risk factors associated with breast cancer.
Red alert
It’s not just the food itself that can affect the risk of breast cancer; how you cook the food can influence its role in this disease. Recent evidence linking the consumption of barbequed and smoked meats to breast cancer was provided by a large study published in the journal Epidemiology (Steck et al., 2007). This study shows that postmenopausal (but not premenopausal) women with a high lifetime intake of grilled, barbequed and smoked meats have a 47 per cent increased risk of breast cancer. This study also reported that big meat-eaters who also skimp on fruit and vegetables were found to have a massive 74 per cent increase in risk. The authors concluded that these results support the accumulating evidence that the consumption of meats cooked by methods that promote carcinogen formation may increase risk of postmenopausal breast cancer. Why this effect was not seen in premenopausal women is unclear. That said, it would seem prudent for women of all ages to avoid barbequed and smoked meats in order to reduce the risk of breast cancer.
It’s not all gloom and doom though, there are many things you can do to protect yourself against breast cancer and other diseases. For example: stopping smoking, losing weight, exercising more and cutting down on alcohol. Changing the way you eat is vital and in your control.
Fruit and vegetables
Fruit and vegetables can protect against breast cancer through a variety of mechanisms involving their antioxidant, fibre and other nutritional content (Michels et al., 2007). Indeed the evidence linking fruit and vegetable consumption to a reduced risk of breast cancer is accumulating.
A large-scale US survey published in the Journal of the National Cancer Institute called the Nurses’ Health Study, reported a negative association between premenopausal breast cancer and women consuming five or more vegetables per day compared with those consuming less than two vegetables per day (Zhang et al., 1999). In other words, premenopausal women who ate the most vegetables had a lower risk of developing breast cancer than those who ate the least. The authors concluded that the consumption of fruits and vegetables high in specific carotenoids (nutrients found in brightly coloured vegetables and fruits such as carrots, sweet potatoes, spinach, kale, spring greens and tomatoes) may reduce breast cancer risk among premenopausal women. This, they noted, applied particularly to those who were at a higher risk because of a family history of breast cancer or consumption of alcohol.
Another study looking at the role of diet and breast cancer risk in 8,984 Italian women over nineand-a-half years follow-up (the length of time that passes between the start of the study when information is collected and the end of the study when the participant’s health status is assessed) recorded 207 cases of breast cancer (Sieri et al. 2004). This study identified four dietary patterns among the participants: salad vegetables (mainly consisting of raw vegetables and olive oil); Western (mainly consisting of potatoes, red meat, eggs and butter); canteen (pasta and tomato sauce); and prudent (cooked vegetables, pulses and fish, with little or no wines and spirits). Results showed that only the salad vegetables dietary pattern was associated with significantly lower (34 to 35 per cent) breast cancer incidence. Women with body mass index of less than 25 had an even greater risk reduction (50 per cent). Whereas for women with body mass index above 25, the salad vegetable dietary pattern had no protective effect. The authors suggest this latter finding may reflect how leaner women might be more health conscious and how overweight women may be dieting and so consuming more vegetables than they would normally. These findings provide compelling evidence that a healthy diet, containing plenty of salad vegetables, can protect against breast cancer.
Three years later, a further 31 breast cancer cases had occurred among this cohort and new analysis revealed that that the salad vegetables dietary pattern offered a significant amount of protection (75 per cent lower), against a specific subtype (HER-2-positive) of breast cancer (Sant et al., 2007). HER-2positive breast cancers have higher than normal levels of a protein known as HER-2 on the surface of the cancer cells. This protein encourages the cancer cells to grow which is why HER-2-positive cancers tend to grow faster than those that are HER-2 negative. Approximately 20 to 25 per cent of breast cancer patients are described as having HER-2-positive breast cancer (Breast Cancer Care, 2007a).
Taken together, this research suggests that fruit and vegetables play an important role in protecting against breast cancer. This is certainly an area that warrants further investigation.
Folic acid
Folic acid (folate) is an important B vitamin necessary for the production of red blood cells and the synthesis of deoxyribonucleic acid (DNA – the blueprint for life). It is also required for the normal methylation of DNA. This refers to the ‘flagging’ of genes with a methyl group (a group of carbon and hydrogen atoms) that is used to mark out specific genes for expression (activation). This process is essential for normal growth, development and function. If the methylation process is disrupted, unregulated gene expression may lead to uncontrolled cell division or cancer.
The deficiency of certain vitamins has been said to mimic the effects of radiation damage to DNA. Indeed a low intake of folic acid could increase the risk of several cancers, including breast cancer, whereas a sufficient intake of folic acid and related B vitamins may protect against this disease (Wang et al., 2006).
Several studies show how dietary folic acid might protect against breast cancer, particularly among women who are at an increased risk as a result of high alcohol consumption. A prospective study of 17,447 Anglo-Australian women (among whom there were 537 cases of invasive breast cancer) investigated the effects of dietary folic acid on the relation between alcohol consumption and breast cancer risk (Baglietto et al., 2005). Results showed that women who had a high alcohol consumption coupled to a low intake of folic acid had an increased risk of breast cancer, but those with a high alcohol consumption and a moderate to high intake of folic acid had no increased risk. It was concluded that an adequate dietary intake of folic acid might protect against the increased risk of breast cancer associated with high alcohol consumption.
Folic acid is found in many foods including broccoli, Brussels sprouts, asparagus, peas, chick peas and brown rice. Other useful sources include fortified breakfast cereals, wholegrain bread and certain fruits (oranges and bananas).
Fibre
Fibre has been hypothesised to reduce breast cancer risk through a number of mechanisms including the inhibition of oestrogen reabsorption back into the bloodstream, the inhibition of human oestrogen synthetase (an enzyme that we use to make oestrogen) leading to a reduction in oestrogen production and finally, a reduction in the levels of steroid hormones called androgens which influence levels of oestrogens and proliferation of breast tissue or by some mechanism involving insulin and IGFs (Cade et al., 2007). As stated above, IGF-1 is associated with an increased breast cancer risk and IGF-1 levels are influenced by diet.
Fibre intake and breast cancer incidence was investigated among a large group of women called the UK Women’s Cohort, which included high number of vegetarians to allow for meaningful comparisons with fish and meat-eaters (Cade et al., 2007). This study showed that in premenopausal, but not postmenopausal women, those who ate 30 grams of fibre per day had half the risk of those who ate less than 20 grams. These findings suggest that in premenopausal women, total fibre is protective against breast cancer; in particular, fibre from cereals and possibly fruit. Dietary fibre has been shown to regulate oestrogen levels in the body. The researchers believe that this may explain why the effects of increased fibre intake were only seen in premenopausal women and not in postmenopausal women.
This is the first large prospective study to show a relationship between total fibre intake and risk of premenopausal breast cancer. Previous analysis may have missed such a relationship between fibre and breast cancer by combining premenopausal and postmenopausal results together.
The average person in the UK eats 12 grams of fibre per day. To increase your intake you could choose a high-fibre cereal for breakfast or switch from white bread and pasta to wholemeal and ensure you have at least five portions of fruit and vegetables per day.
Eat your greens!
It is well-documented in the scientific literature that cruciferous vegetables (such as spring greens, broccoli, Brussels sprouts and kale) help protect against cancer. Some of this activity has been attributed to a compound in these vegetables called sulphurophane which has attracted much attention since its discovery in 1992. The mechanism underlying this compound’s anti-cancer activity has remained unclear until 2004. In a study published in the US Journal of Nutrition scientists proposed just how this compound may offer protection (Jackson and Singletary, 2004). Keith Singletary and colleagues at the University of Illinois exposed malignant human breast cells to sulphurophane in the laboratory and saw that it inhibited cell division and DNA synthesis in the cancer cells. In other words, this compound inhibited the growth of cancer cells, but not normal cells.
Singletary suggests that sulphurophane works by disrupting cellular components called microtubules. These are long slender cylinders of a protein called tubulin that are required for the normal separation of the duplicated chromosomes during cell division. If this process is disrupted then cells cannot divide. These findings may be useful in developing new treatments for breast cancer. In the meantime, including a wide range of vegetables in the diet, including the cruciferous varieties, could offer immediate benefit.
The soya connection
There is strong evidence that soya intake during adolescence may reduce the risk of breast cancer later in life. The Shanghai Breast Cancer Study investigated the role of soya in breast cancer by looking at the dietary history of 1,459 breast cancer cases and 1,556 age-matched controls in China (Shu et al., 2001). This study found that women who consumed the most soya as teenagers had half the risk of breast cancer as adults. This inverse association was observed for each of the soya foods examined (tofu, soya milk and other soya products) and existed for both premenopausal and postmenopausal women. The authors of this study suggest that a substantial difference in breast cancer incidence between Asian and Caucasian women and increasing breast cancer incidence among Asian-Americans may be explained, at least in part, by soya food intake during adolescence. They state that their study emphasises the importance of initiating cancer intervention programs early in life.
A year later, scientists investigating the link between adolescent soya intake and breast cancer asked over 1,000 Asian-American women (including 501 breast cancer patients) how often they ate soya foods such as tofu, soya milk and miso. This study also found that women who consumed soya at least once a week during adolescence had a significantly reduced risk of breast cancer (Wu et al., 2002).
Results showed that those who were high soya consumers as both adolescents and adults had a 47 per cent reduction in risk of breast cancer. Those who ate little soya as adults but had eaten it regularly during adolescence showed a 23 per cent reduction in risk. Women who were low consumers during adolescence and high consumers during adulthood showed little reduction in risk. These studies suggest that high soya intake during adolescence reduces breast cancer risk and the risk continues to fall if people continue to eat soya as an adult.
In an attempt to draw the scientific evidence together, a team lead by Professor Bruce J. Trock from the Johns Hopkins School of Medicine in Baltimore, MD, performed a review of 18 studies on soya exposure and breast cancer risk published between 1978 and 2004 (Trock et al., 2006). Results showed a modest association between a high soya intake and a reduced breast cancer risk. The authors warn that this result should be interpreted with caution and that recommendations for high-dose isoflavone supplementation to prevent breast cancer or prevent its recurrence would be premature. However, they do state that there is no evidence to suggest that consumption of soya foods, in amounts consistent with an Asian diet, is detrimental to breast health, and go on to say that such a diet is likely to confer benefits to other aspects of health. In other words, the research looks promising but more evidence is needed.
The low rate of breast cancer in Japan and the high rate of survival amongst those who are affected is often used to promote soya foods as being beneficial – or at least, not being harmful – for breast cancer patients. Some researchers, however, are cautious and think that even the small oestrogenlike effect of soya foods may be detrimental for women who have gone through the menopause, whose natural oestrogen levels have dropped and who have been diagnosed with oestrogen-receptor positive (hormone sensitive) breast cancer (PCRM, 2002).
Their concern is that the weak oestrogen activity of soya isoflavones may stimulate the growth of tumours which are sensitive to oestrogen. This is not a concern for premenopausal women, who have much higher levels of oestrogens which are many times more potent than phytoestrogens.
These concerns are based largely on the results of a small number of in vitro (test tube) and animal studies but as these have produced mixed results their relevance to human breast cancer patients remains unclear.
To date there have only been two human studies on this subject, the findings of which were also unclear (Petrakis et al., 1996; Hargreaves et al., 1999). The cautious approach would be for postmenopausal women at risk of breast cancer to limit the amount of soya products they eat to three or four a week.
On the whole, the evidence suggests that consuming moderate amounts of soya foods is much more likely to benefit health rather than harm it, both in terms of breast cancer risk and other chronic diseases.
For more information on foods that can help fight cancer see the easy-to-read colourful guide A Fighting Chance. A guide to healthy eating to help prevent and overcome breast cancer available clicking here. This guide summarises the key findings this report and provides vital information on a range of cancer-busting foods. It also includes a seven-day meal plan with inspiring healthy recipes such as our ever-popular Tortilla Wraps with Mango Salsa, Quinoa Superbowl Salad and the fabulous Summer Berry Compote.
Identifying specific foods (or components of them) that can increase the risk of, or even cause, certain diseases, is notoriously difficult. There are many problems associated with trying to tease out the links between diet and disease. For example, most diet and breast cancer risk studies have been conducted in industrialised countries (North America, Europe and Japan). Comparing the diets between industrialised and developing countries rather than within them could offer more insight as the diets between these countries tend to vary more. This may permit a better comparison, for example, of a plant-based diet versus an animal food-based diet or a soya versus non-soya diet.
Another problem is that self-reported diets (food diaries and food frequency questionnaires) are often assessed with considerable measurement error. Furthermore, most studies tend to focus on the diet consumed as an adult, whereas strong evidence suggests dietary influences before adulthood can affect breast cancer risk later in life. For example, research shows that a high soya intake during adolescence can reduce the risk of breast cancer later in life. The average follow-up time may be too short to determine significant conclusions. The effect of diet on different sub-types of cancer such as: oestrogen receptor-positive, progesterone receptor-positive, genetic (due to faulty genes), epigenetic (not due to genes) warrants further investigation. For example, salad vegetables have been shown to lower the risk of HER-2 positive breast cancer. The positive effect of some foods may be masked by the negative effects of others. For example, in the Shanghai Study the authors suggest that the potential positive effect of vegetables and soya foods may have been countered by the negative effects of fish in the diet. Furthermore, the effects of specific diets (such as macrobiotic, organic, wholefood, raw food or vegan) have not been sufficiently studied.
The supposed health benefits of meat and dairy foods have been vigorously promoted by the meat and dairy industries for decades. For example, the idea that meat is essential for iron and protein is deep-rooted and is often used to pressure would-be veggies back to the butchers. The reality is that we do not need saturated animal fat, animal protein or cholesterol. We do not need the trans fatty acids in processed foods. We do not need the amount of salt and sugar we consume. We do however need to move towards a plant-based, wholegrain diet containing a wide range of fruits, vegetables, grains, pulses, nuts and seeds for the nutrients that will promote a long and healthy life.
These, of course, are the same foods which contain protection against disease in the form of antioxidants and fibre. What is killing the Western world are the degenerative diseases associated with affluence. It is clear that the same diet that is good for preventing breast cancer is also good for preventing heart disease, obesity, diabetes and so on.
The milk debate deserves a special mention here as the notion that cow’s milk is a natural and healthy drink for humans is so deeply entrenched in the British psyche, yet the evidence suggests milk may be doing us more harm than good. Of course we need calcium for our bones and teeth
(and blood clotting, muscle function and regulating heart rhythm). But despite the dairy industry’s powerful marketing machine, more and more people are beginning to wonder if cow’s milk really is the best source of calcium. It certainly is not for most of the world’s people. Claims that dairy is best carry strong overtones of cultural imperialism and simply ignore the 70 per cent of the global population who obtain their calcium from other sources – people such as the Japanese who traditionally have consumed no dairy yet have far better health than British people and live considerably longer.
Milk has been part of the human diet for less than 6,000 years; this is very recent in evolutionary terms. It is not just that most people don’t drink it; they cannot because their bodies will not tolerate it. Up to 100 per cent of some ethnic groups are lactose intolerant. In global terms lactose intolerance is very common, occurring in around 90-100 per cent of Asians, 65-70 per cent of Africans, but just 10 per cent of Caucasians (Robbins, 2001). This suggests that the health claims made for milk owe more to marketing than science.
The dairy industry has spent many years and many millions of pounds promoting the notion that cow’s milk is good for us through expensive advertising campaigns such as the ‘White Stuff’. Now, because of an increasing body of evidence, there are signs of a growing realisation that milk is neither natural nor healthy. In fact, research is moving in the opposite direction now, showing that the more dairy and animal protein that is consumed, the higher the incidence of osteoporosis and other diseases.
The rate at which some cancers are increasing is a huge matter of concern. When Professor Jane Plant wrote the first edition of Your Life in Your Hands in 2000, one in 10 UK women were affected by the disease. Now, in 2007, one in nine women are expected to develop breast cancer at some point in their lives! Since 1971, the incidence of breast cancer in the UK has increased by 80 per cent. These figures should be shouted from the rooftops! An increasing number of researchers are in no doubt that cow’s milk and dairy foods are responsible.
A point that is consistently overlooked is that two-thirds of the UK’s milk comes from pregnant cows and as every mum knows, hormone levels during pregnancy can rise dramatically. This is no laughing matter as breast, prostate, ovarian and colorectal cancer are all implicated. These cancers and the so-called diseases of affluence, such as diabetes, obesity, heart disease and even osteoporosis, occur increasingly in the countries that consume the most dairy products. It is not rocket science… cow’s milk and dairy products cause disease.
The conclusions of this report are drawn from a huge body of research from academic institutions all around the world. While the majority of this work was performed in an academic environment (involving clinical trials or statistical analysis), some is of a more personal nature. Professor Jane Plant’s spirit and courage in overcoming breast cancer through the elimination of all dairy could not fail to inspire the increasing number of women who are affected by this disease. Plant did not set out to promote one type of diet above another but as a scientist (geochemist) she took an analytical approach to the problem of breast cancer and ultimately found the solution: a dairy-free diet.
In summary, this report provides a compelling argument that the consumption of animal-based foods is linked to the development of breast cancer. The combined findings of over 50 scientific papers from reputable peer-reviewed journals such as the British Medical Journal and the Lancet leave no doubt that diet is linked to breast cancer risk. Taken in their entirety, they indicate a causative role for red meat, animal fat and dairy foods. This report provides a vital source of information for health professionals, enabling them to make better-informed choices in recommending dietary changes to breast cancer patients and women considered to be at risk of this disease.
The official approach to the causes of breast cancer (and other so-called diseases of affluence) remains extremely equivocal and dietary advice seems to be based far more on not upsetting particular vested interests than improving the public’s health. As a consequence, the incidence of these diseases continues to rise remorselessly because public health policy is aimed, almost exclusively, at treatment rather than prevention.
Only when prevention becomes the priority will the avoidance of animal products be seen as central to improving public health. The World Health Organisation believes that the only way we can improve our health is through informed opinion and active co-operation. We agree! As a science-based health charity, Viva!Health provides unbiased information on which people can make informed choices. We monitor and interpret scientific research on diet and health and communicate those findings to the public, health professionals, schools and food manufacturers. Importantly, we have no commercial or vested interests and offer a vital – and what sometimes feels like a solitary – source of accurate and unbiased information.
So it is up to individual members of the public and independent-minded health professionals to find out what they can about diet and heath. Meanwhile, government health policy continues unchanged, like Nero fiddling while Rome burns.
Be a bosom buddy…
Tell your friends about it by giving them our easy-to-readguide with seven-day meal plan and recipes.
A Fighting Chance: a Guide to Healthy Eating to Help Prevent and Overcome Breast Cancer. £1.90 (plus p&p) available from Viva!
This easy-to-read colourful guide summarises the key findings of our scientific report on breast cancer and provides vital information on which foods can help fight cancer. Also includes a seven-day meal plan with inspiring healthy recipes including our ever-popular Tortilla Wraps with Mango Salsa, Quinoa Superbowl Salad and the fabulous Summer Berry Compote.
Order by post, by phone or online. Viva!Health, 8 York Court, Wilder Street, Bristol BS2 8QH
T: 0117 970 5190 (Mon-Fri 9am-6pm) W: www.viva.org.uk
About Viva! Health
Viva!Health is a science-based health and nutrition charity which monitors and interprets the growing body of research linking diet and health. Viva!Health helps the public, health professionals and the food industry make informed choices about diet by providing accurate information and advice about healthy eating. Viva!Health also runs health and education campaigns, presents school talks, cookery demonstrations, produces a magazine, Viva!Life!, and a wide variety of materials, runs the Vegan Recipe Club and answers nutritional queries from the public. The majority of diseases that kill most of us prematurely can be prevented by consuming a plant-based diet – Viva!Health explains why and provides information and advice about healthy eating.







